
CAS number: 134523-03-8
Molecular formula: C66H68CaF2N4O10
molecular weight: 1155.34
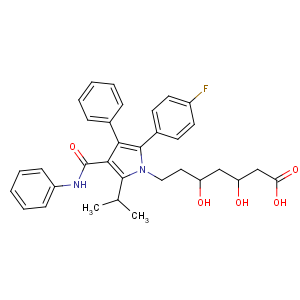
Chemical structure:
| SR.NO | TEST | SPECIFICATION | RESULT | ||||||
| 1 | DESCRIPTION | White to off white crystalline powder | White crystalline powder | ||||||
| 2 | Solubility | Freely Soluble in methanol , slightly soluble in alcohol, very slightly soluble in distilled water, in Ph 7.4 phosphate buffer and in acetonitrile, insoluble in aqueous solution of pH 4 and below. | Complies | ||||||
| 3 | Identification | A. By IR :The IR spectrum of sample should be concordant with the IR spectrum of standard. | Complies | ||||||
| B. Calcium : The sample solution exhibits a significant absorption at the calcium emission line at 422.7 nm. | Complies | ||||||||
| 4 | Organic impurities by HPLC | Atorvastatin related Compound –A : Not more than 0.3% | 0.09% | ||||||
| Atorvastatin related Compound –B : Not more than 0.3% | Not detected | ||||||||
| Atorvastatin related Compound –C : Not more than 0.3% | 0.03% | ||||||||
| Atorvastatin related Compound –D : Not more than 0.2% | Not detected | ||||||||
| Any other individual impurities : Not more than 0.1% | 0.10% | ||||||||
| Total impurities: Not more than 1.0% | 0.34% | ||||||||
| 5 | Enantiomeric purity by HPLC | Atorvastatin related Compound –E : Not more than 0.3% | 0.04% | ||||||
| 6 | Heavy Metals | Not more than 20 ppm | Less than 20 ppm | ||||||
| 7 | Water content by KFR | Not less than 3.5% w/w and not more than 5.5% w/w | 4.26% w/w | ||||||
| 8 | Assay by HPLC | Not less than 98.0% w/w and not more than 102.0% w/w (on anhydrous basis ) | 99.27% w/w | ||||||
| 9 | Residual Solvents by HS- GC | Metanol: Not more than : 3000 ppm | 29 ppm | ||||||
| Isopropyl alcohol: Not more than : 5000 ppm | 11 ppm | ||||||||
| Acetone: Not more than : 5000 ppm | 06 ppm | ||||||||
| Cyclohexane: Not more than 3880 ppm | 21 ppm | ||||||||