
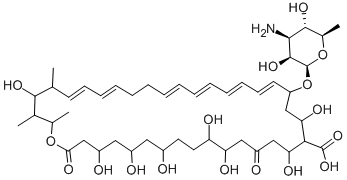
CAS number: 1400-61-9
Molecular formula: C47H75NO17
molecular weight: 926.09
Chemical structure:
| Sr No. | Test | Specification | Result |
| 1 | Appearance | Yellow or slightly brownish powder, hygroscopic , yellow to light tan powder, having an odor suggestive of cereals. Is hygroscopic, and is affected by long exposure to light ,heat, and air | Yellow powder, hygroscopic, having an odor suggestive of cereals |
| 2 | Solubility | Freely soluble in dimethylforamide and in dimethylsulfoxide sparingly to slightly soluble in methanol, in n-propyl alcohol and in n-butyl alcohol practically insoluble in water and in alcohol ,insoluble in chloroform and in ether | Corresponds |
| 3 | Identification A 230/ A280 A 291/ A205 A 319/ A305 Identification ,ultraviolet Absorption A230/A279 (sh) | Absorption maxima at 230 nm ,291 nm,305 nm ,319 nm And a shoulder at 280 nm 0.83 to 1.25 0.61 to 0.73 0.83 to 0.96 0.90 to 1.25 | Corresponds 1.06 0.64 0.9 1.1 |
| Identification B | Complies with requirements | Corresponds | |
| Identification C | Complies with requirements | Corresponds | |
| Identification D | Complies with requirements | Corresponds | |
| Identification E | Complies with requirements | Corresponds | |
| Absorbance at 305 nm | Not less than 0.60 | 0.823 | |
| 4 | Loss on drying –USP 39 | Not more than 5.0 % | 1.35 |
| BP 2016, Eur.Ph 9th edition | 3.67 | ||
| 5 | Heavy metals | Not more than 20 ppm | < 20 |
| 6 | Sulphated ash | Not more than 3.5 % | 0.67 |
| 7 | Assay – USP 39 | Not less than 4400 USP Nystatin Units/mg or where intended for use in the extemporaneous preparation of oral suspensions,not less than 5000 USP Nystatin Units/mg | 6199 |
| BP 2016 / Eur.Ph.9th edition | Minimum 4400 IU/mg (dried substance ) and minimum 5000 IU/mg (dried substance ) if intended for oral administration | 6435 | |
| 8 | Abnormal toxicity | In conformity with BP 2016 and Eur.Ph. 9th edition (2.6.9) | Corresponds |
| 9 | Suspendibility | Not less than 90.0 % | 93.2 |
| 10 | Crystallinity | It reveals birefringence | Corresponds |
| 11 | pH ( 3.0 % aqueous suspension ) | Between 6.0 and 8.0 | 7.51 |
| 12 | Composition USP 39 – Nystatin A1 Any other individual component | Minimum 85.0 % | 90.57 |
| Maximum 4.0 % | 1.17 | ||
| BP 2016/ Eur. Ph. 9th edition -Nystatin A1 Any other compound | Minimum 85.0 % | 91.26 | |
| Maximum 4.0 % | 1.16 | ||
| 13 | Microbiological quality TAMC,CFU/g TYMC,CFU/g | Not more than 103 | 30 |
| Not more than 102 | < 1 | ||
| Specified Micro-organisms Bile –tolerant gram-negative bacteria/g Escherichia coli/g Salmonella/g Pseudomonas aeruginosal/g Staphylococcus aureus /g | Absent | Absent | |
| 14 | Absent | Absent | |
| Absent | Absent | ||
| Absent | Absent | ||
| Absent | Absent | ||
| 15 | Residual solvents Methanol Acetone | Not more than 0.3 % | NA |
| Not more than 0.5 % | NA | ||
| 16 | Partical Size | Not more than 45 µm | 35.73 |